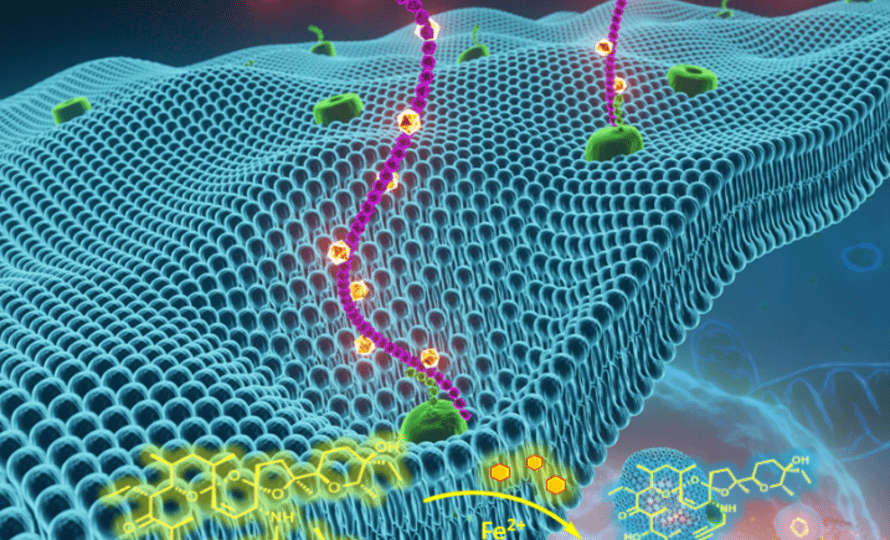
Published in the Journal of the American Chemical Society, these results use iron chemistry and pave the way for a new class of antitumor drugs.
Pancreatic cancers have a very poor prognosis, and there is an urgent need for new therapeutic options. Raphaël Rodriguez, CNRS research director in the Cellular and Chemical Biology unit (Institut Curie, Inserm, CNRS), and his team are dissecting the iron chemistry involved in tumors and the metastatic process, to invent new therapeutic strategies. Researchers conducted a specific study of pancreatic tumor cells. Decoding.
Using ferroptosis to eliminate pancreatic tumor cells
Iron is involved in the regulation of many cell processes including some that allow cells to become metastatic and resistant to conventional antitumor treatments. Due to the presence of iron, however, these same cells have increased sensitivity to ferroptosis: cell death caused by the uncontrolled formation of certain lethal compounds (lipid peroxides), which form in a specific cell compartment (lysosome). This sensitivity led researchers to implement a strategy to specifically target this chemical pathway.
This study again demonstrates that chemical processes represent a means to to promote death of cancer cells, in this case, pancreatic cells
Asserts Raphaël Rodriguez.
Thanks to the chimeric small molecules that we developed, our therapeutic window is expanding: we can specifically target the tumor cells that are most harmful for the body, while sparing healthy cells. If we can continue the research to clinical stages, the prospects are promising.
A new class of anticancer “prodrug”
Raphaël Rodriguez’ team was thus able to synthesize a series of chimeric small molecules, called prodrugs. Researchers demonstrated that these next-generation molecules accumulate in the lysosomes of persister cells. Due to the effect of iron, bioactive species are released and, in various models, cause cell death via ferroptosis. Iron thus acts as a chemical activator and the biological target. This research also shows that it is possible to induce ferroptosis by directly manipulating iron, particularly the lysosomal pool, a new concept of ‘active ferroptosis’.
Scientists evaluated and demonstrated the action of these drugs on five separate cells lines, involving pancreatic duct adenocarcinoma and patient-derived organoids. The results reveal that the prodrugs tested are effective for eliminating certain derived pancreatic cancer cells, resistant to conventional drugs. This research paves the way for the development of new anticancer strategies involving lysosomal iron and active ferroptosis.
As a co-author on this paper, I can’t resist a nod to history by acknowledging that our illustrious founder Marie Curie had published her work in this same journal 100 years ago
Proclaims Alain Puisieux, director of Institut Curie Research Center and study co-author
These results bolster the chosen strategy of targeting iron in tumor cells. It is a concrete proof of the potential of chemical biology, a discipline that combines the principles of physical chemistry with knowledge in biology, to have an impact on medicine. For this reason, at Institut Curie Research Center, we chose to develop this expertise, which promises an exciting future, in terms of therapeutic solutions in oncology.
| Reference:
Iron-Sensitive Prodrugs That Trigger Active Ferroptosis in Drug-Tolerant Pancreatic Cancer Cells. Michał Antoszczak, Sebastian Müller, Tatiana Cañeque, Ludovic Colombeau, Nelson Dusetti, Patricia Santofimia-Castaño, Christine Gaillet, Alain Puisieux, Juan Lucio Iovanna, and Raphaël Rodriguez. Journal of the American Chemical Society. https://pubs.acs.org/doi/10.1021/jacs.2c03973. |